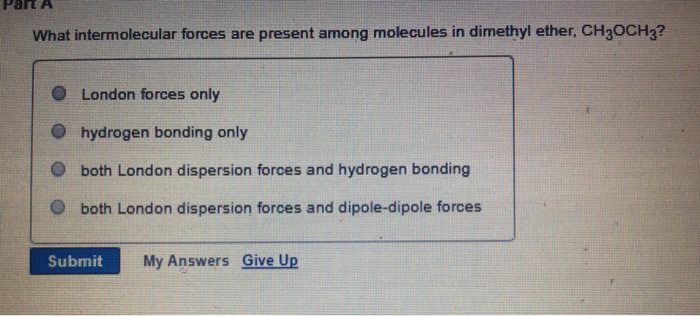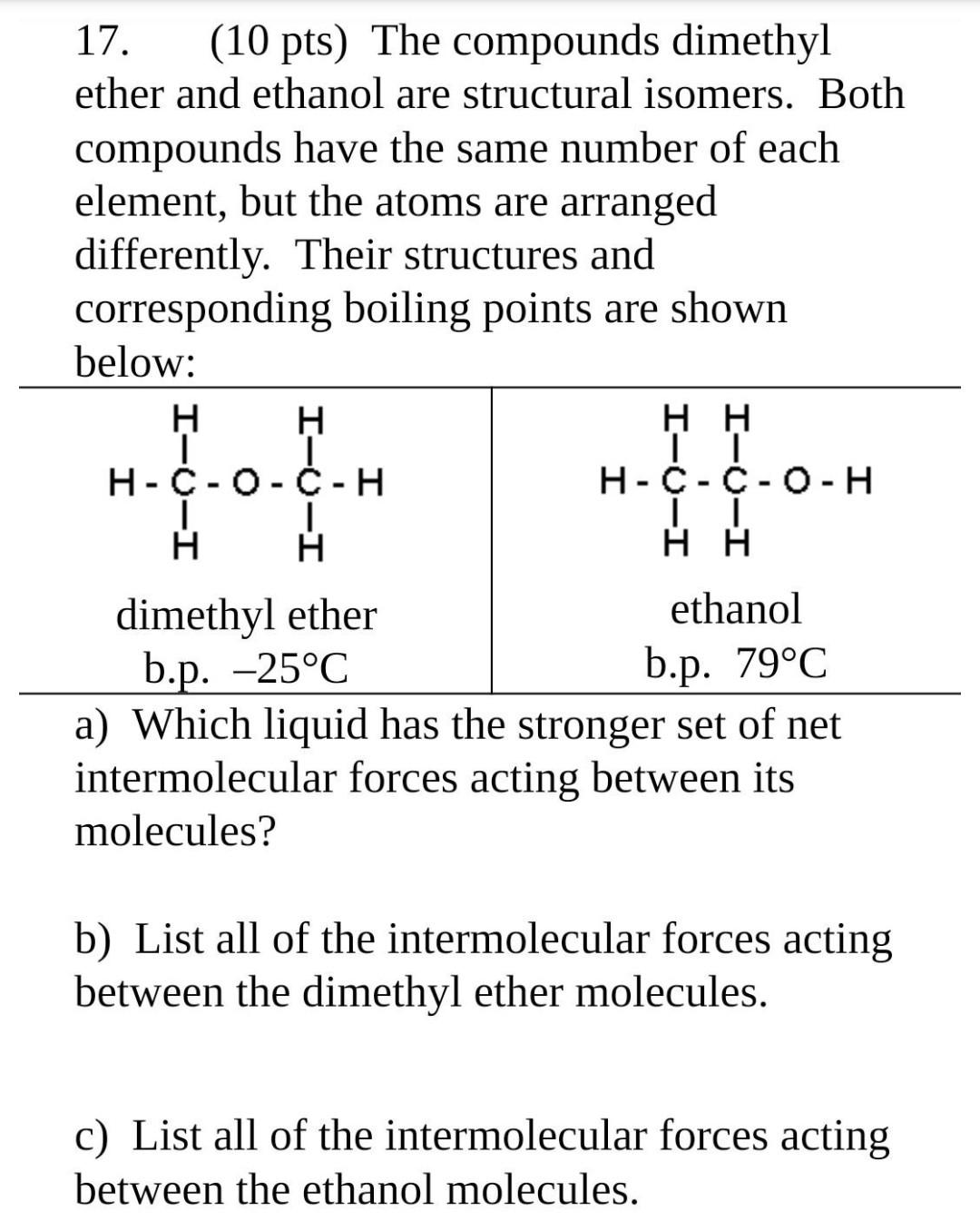
SOLVED: In regards to intermolecular forces, why does dimethyl ester CH3OCH3 boil at -25 degrees and ethanol CH3CH2OH boil at 79 degrees?

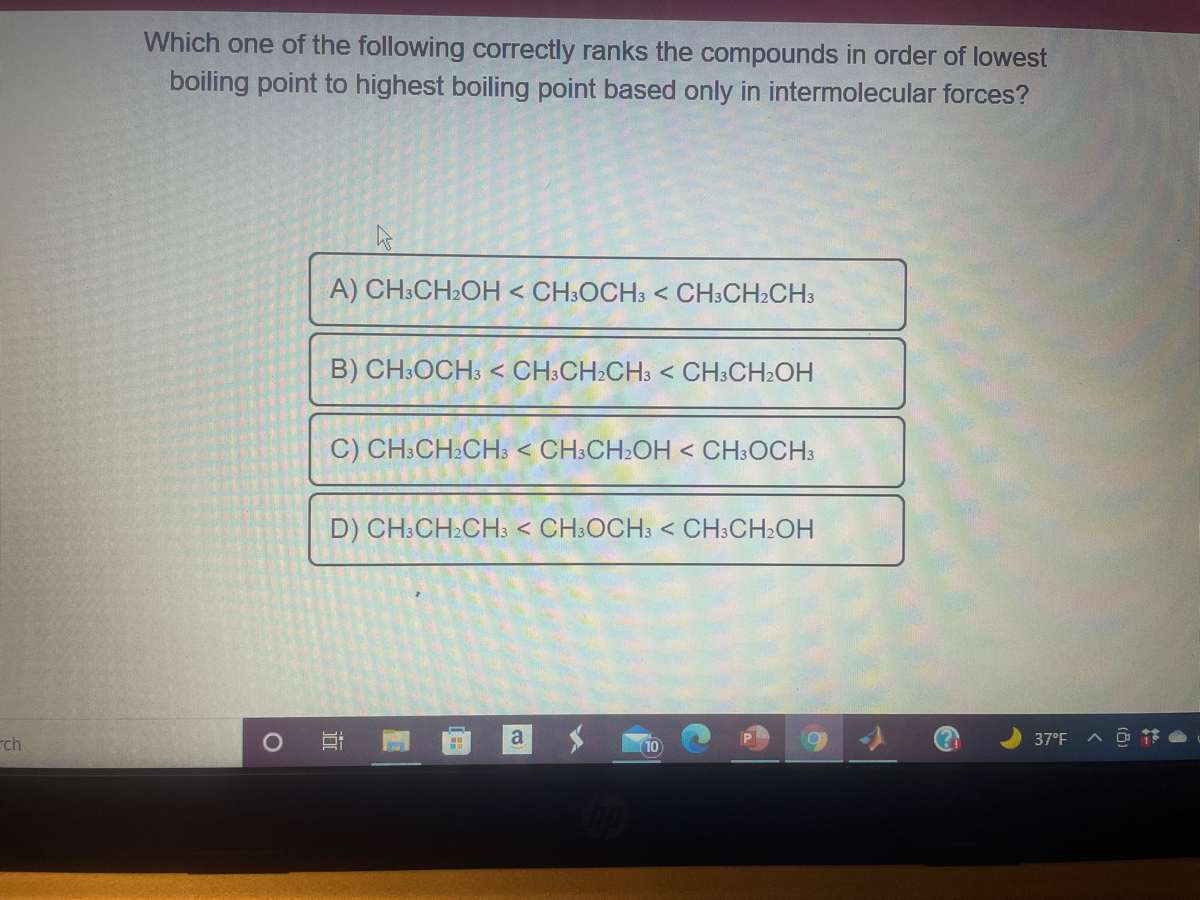
SOLVED:Pick the compound with the highest boiling point in each pair. Explain your reasoning. a. CH3OH or CH3SH b. CH3OCH3 or CH3CH2OH c. CH4 or CH3CH3

Homework 6 - 1. Intermolecular Forces T/F Decide if the following statements regarding Intermolecular forces are True or False. True: NH3 will have a | Course Hero

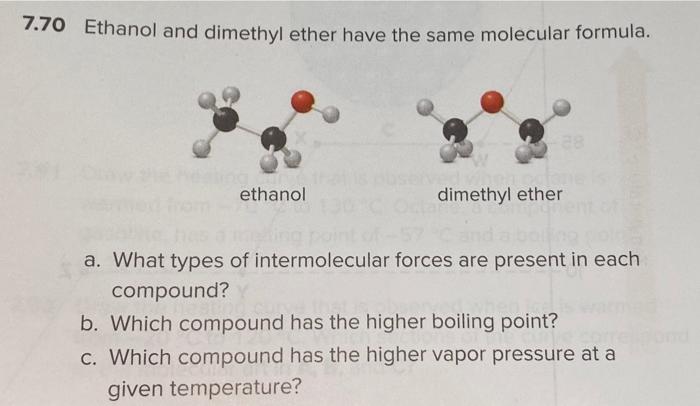
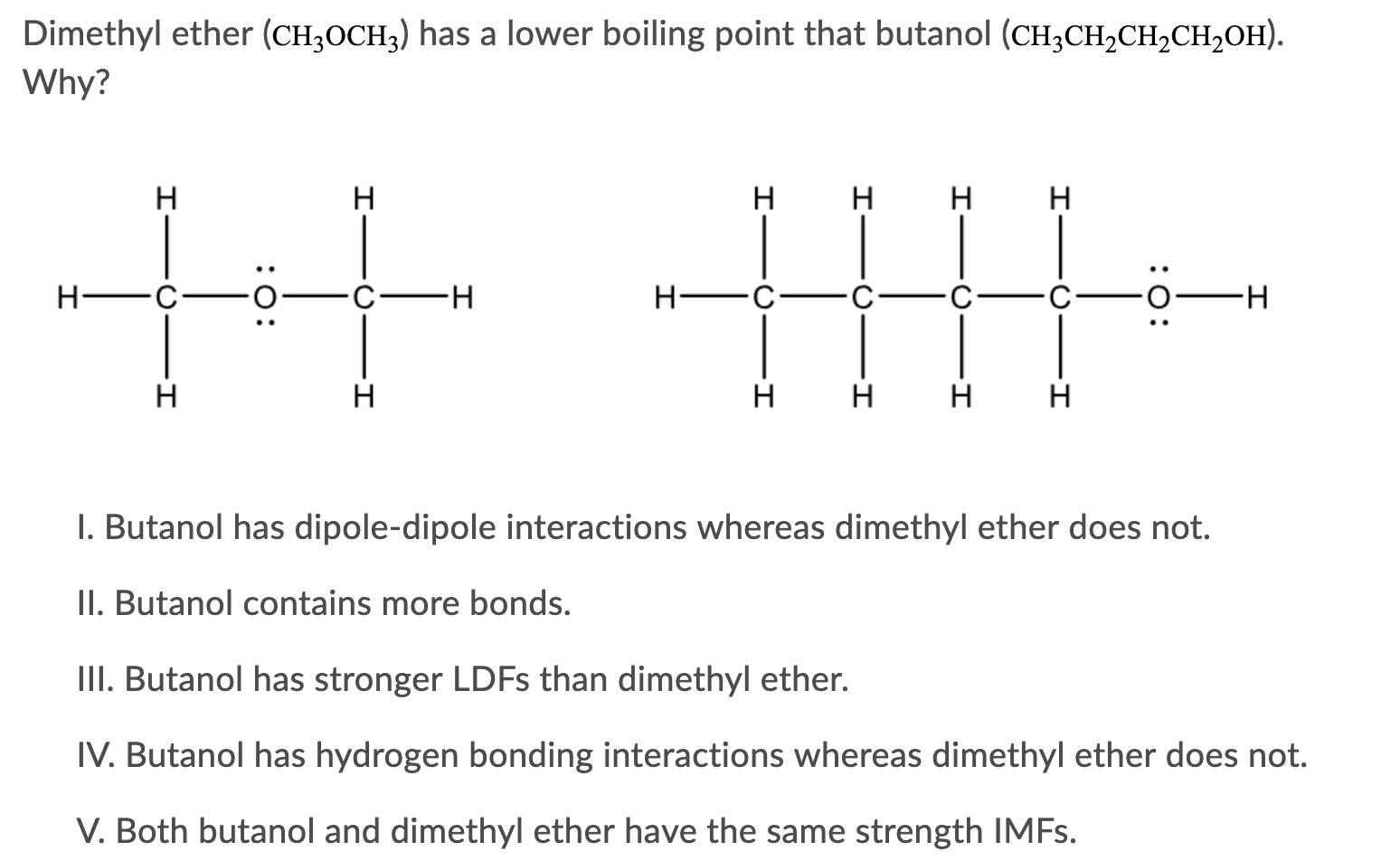
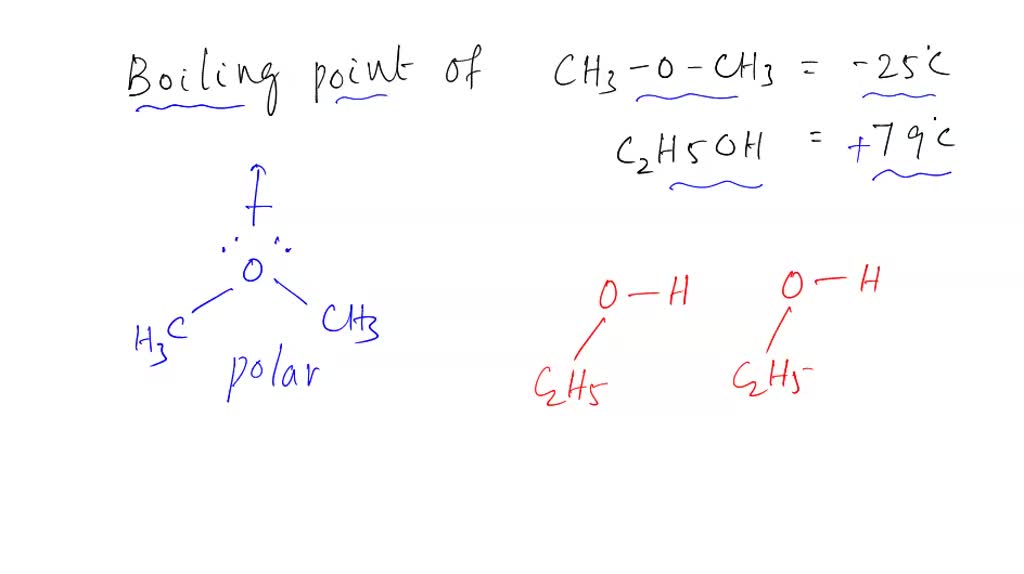
SOLVED: Dimethyl ether (CH3OCH3) has a molar mass of 46 glmol and a boiling point of 248 K. Ethanol (CH:CHzOH) has the same chemical formula and molar mass but a boiling point

Ethyl alcohol and dimethyl ether have the same composition by mass (52% carbon, 13% hydrogen, and 35% oxygen), yet the two have different melting points, boiling points, and solubilities in water. Explain
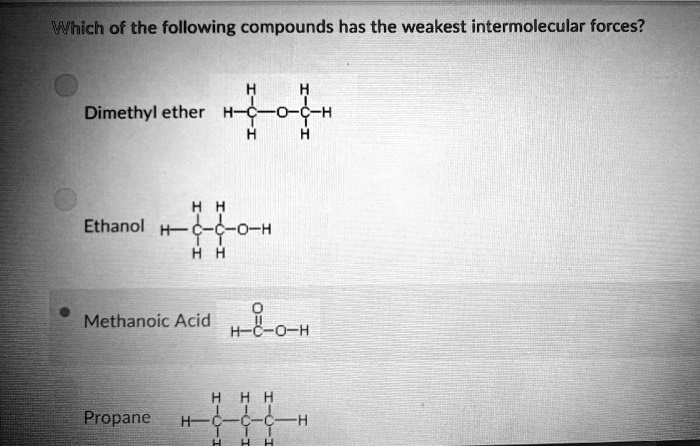
SOLVED: Which of the following compounds has the weakest intermolecular forces? Dimethyl ether C-H Ethanol H 0-H Methanoic Acid H-C-0-H Propane