Which substance likely has a higher boiling point: CF4 or CH2F2? Explain how you know. | Homework.Study.com

Choose the compound that exhibits hydrogen bonding as its strongest intermolecular force. Choose the - Brainly.com

When the substances CCl4, CH4 , and CF4 are arranged in the order of increasing boiling point, what is the correct order?

Relative orientation of the two fragments in the CF 4 , CHF 3 , CH 2 F... | Download Scientific Diagram

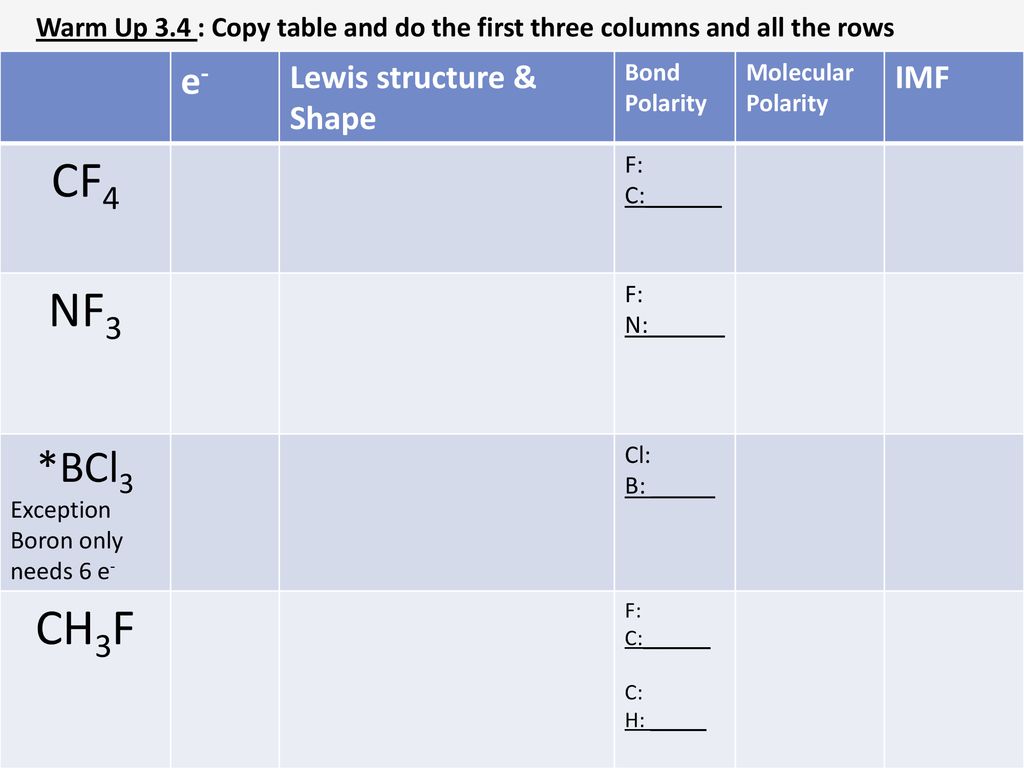
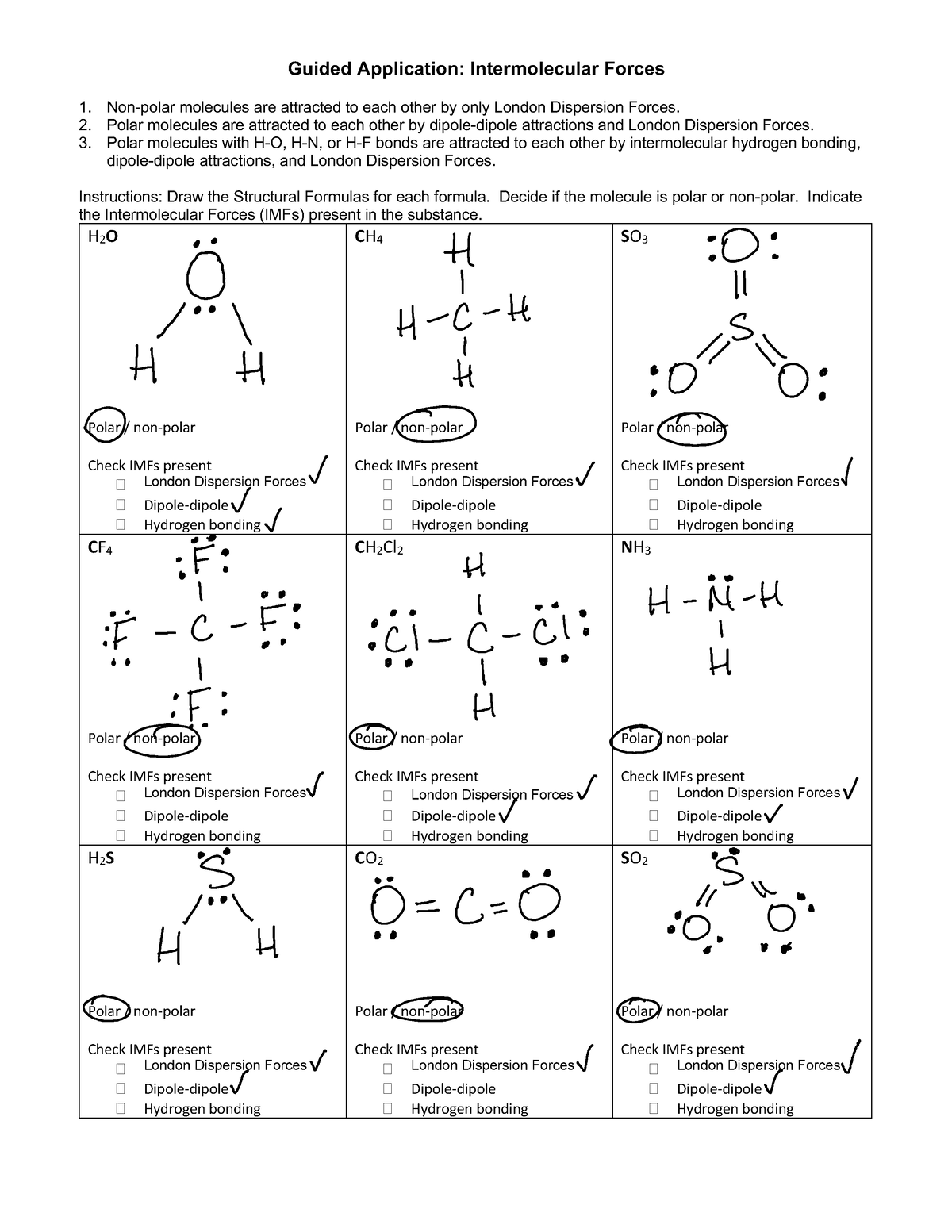
INTERMOLECULAR FORCES (IMF). INTERMOLECULAR FORCES ARE BETWEEN MOLECULES WHEREAS INTRAMOLECULAR FORCES ARE WITHIN THE MOLECULE INTERMOLECULAR FORCES ARE: - ppt download
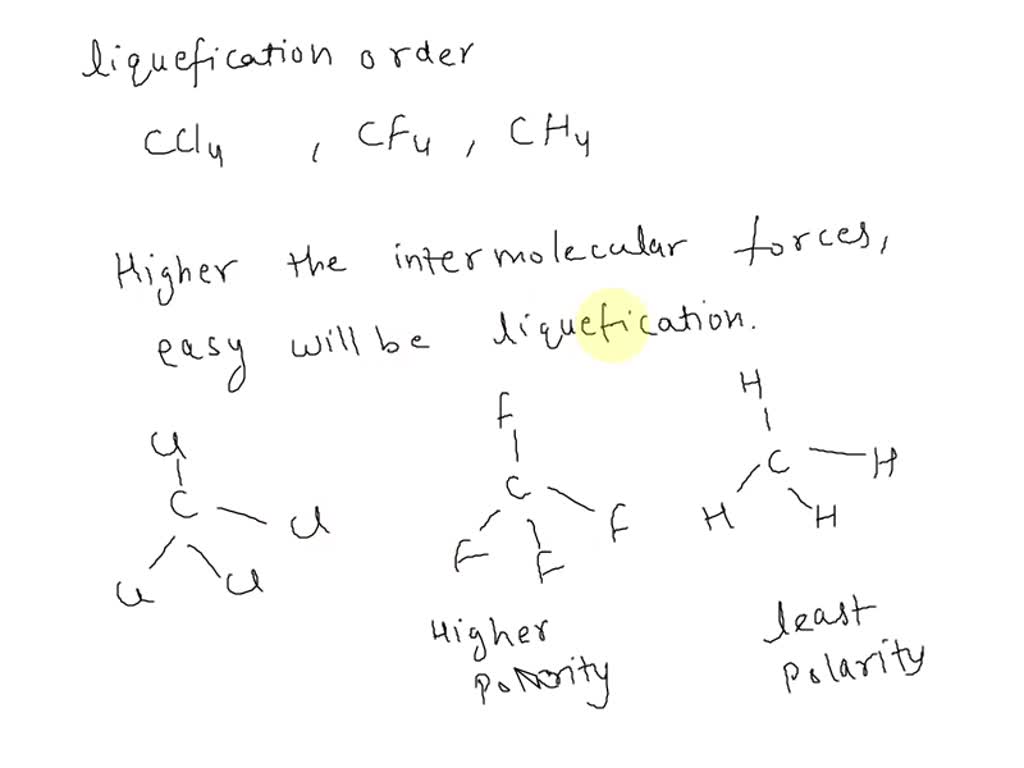
SOLVED: Arrange the following in order of ease of liquefaction; CCl4, CH4, CF4. Explain your ranking.
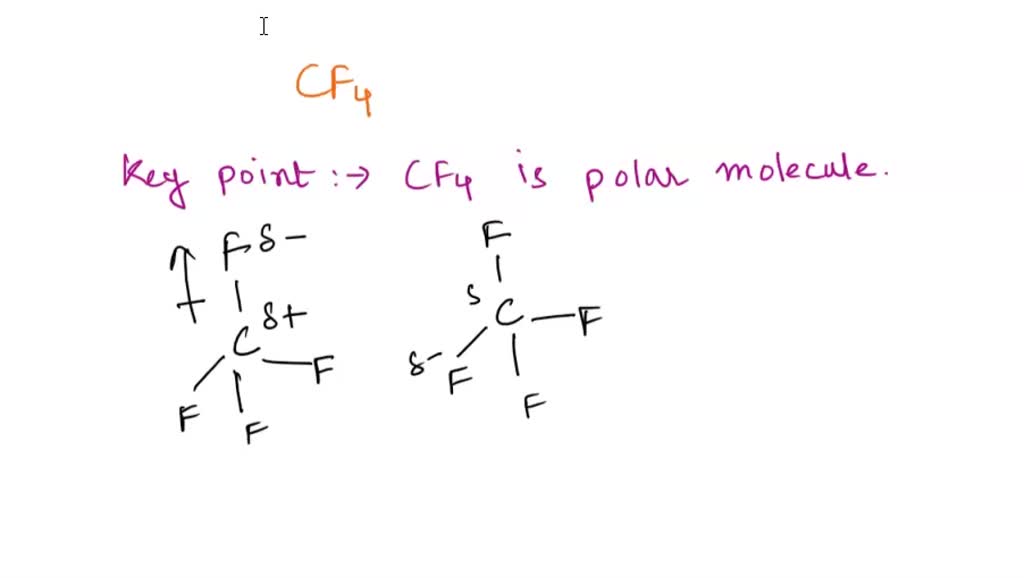
SOLVED: What type is the strongest intermolecular force that must be overcome in converting CF4 from a liquid to a gas? dispersion covalent bond dipole-dipole hydrogen bond